Research Note
Ca-Mg Supplements:
Solubility, Particle Size, Source
Magnesium oxide (MgO) remains a primary source of supplemental Mg in dairy diets, yet calcium-magnesium carbonate supplements also play a key role. Depending on source, Ca-Mg supplements can incorporate bioavailable Mg while supporting rumen alkalinity and calcium homeostasis.
How to distinguish cost-effective Ca-Mg supplements?
At the high-end of cost is CaMg(OH)4 (hydrated dolomitic lime), which requires heat treatment (calcining) of the calcium-magnesium carbonate raw material followed by hydration under steam pressure. This manufactured product offers rapid solubility and absorption in the rumen but typically costs five times the minimally processed alternative — CaMg(CO3)2 (calcium magnesium carbonate).
 “For calcium magnesium carbonate products, the physical source of the raw material is important,” says Iowa State’s Dr. Jesse Goff. “The raw material mineral deposits — limestone and dolomite — are naturally occurring features of diverse, ancient ecosystems. Their crystal structure and impurities vary depending on location, affecting their capacity for hydration and solubility in the rumen. This is also true for magnesium oxide products.”
“For calcium magnesium carbonate products, the physical source of the raw material is important,” says Iowa State’s Dr. Jesse Goff. “The raw material mineral deposits — limestone and dolomite — are naturally occurring features of diverse, ancient ecosystems. Their crystal structure and impurities vary depending on location, affecting their capacity for hydration and solubility in the rumen. This is also true for magnesium oxide products.”
“In fact,” Goff points out, “pure MgO — 60% magnesium and 40% oxygen — is very stable and almost useless in a ration. What makes it bioavailable are imperfections in the crystal structure. The feed ingredient contains 54-56% magnesium, whereby openings in the crystal allow hydration.”
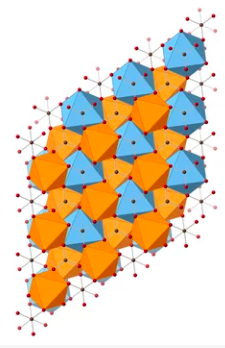
Calcium magnesium carbonate (CaMg[CO3]2) showing pure crystal structure
When it comes to a cost-effective calcium magnesium carbonate product, Goff says, look for:
- Fineness of grind — “avoiding dust, but providing increased particle surface area available for hydration, which increases solubility in the rumen”
- Consistency of grind — relatively narrow particle size distribution
- Quality source of raw material — “which offers ‘hydratable’ Ca-Mg carbonates because failure to become hydrated is usually the stumbling block to bioavailability in less expensive products”
“Magnesium carbonate originating from certain mines or from calcareous marine algae deposits,” Goff says, “can be as reactive as some of the better magnesium oxides on the market. In any case, processing procedures and particle size affect bioavailability of magnesium oxides or carbonates.”
Goff warns about magnesium carbonate byproducts from steel manufacture, where chemical analysis might show high Mg concentration. However, such materials have almost no bioavailability.
More than a decade ago, Goff developed, validated, and published a relatively quick and easy in vitro procedure called the “white vinegar test” for estimating solubility in Mg sources including calcium magnesium carbonates. This test continues to be widely used in the industry and validated by recent studies.
Questions?
Email FeedInsight 4Dairy